There are various elements and compositions around us,aiding in the overall function of the environment. These very
elements are often taken for granted and even overlooked as trivial importance. The ozone layer functions as a protective
canopy to earth and its inhabitants, and unfortunate to say, is one such important element that is overlooked. Throughout the
course of decades, this protective layer has become increasing unstable and depletion of its composition has become
inevitable. The very idea of ozone depletion lies oblivious to the populace beacuse alot of people are unaware of the
importance the ozone and most of all of the threat humanity will incur if this canopy of protection is completely destroyed.
Many people feel that the ozone poses no imminent threat to humanity and that ozone depletion is simply a natural process.
While this is true, in part, circumspect evaluation of the facts will prove that there has been a rather unaccounted decrease in
ozone, especially over the region of Antarctica. This depletion of the ozone layer is not without reason and responsibility. In
essence, for the most part this rapid wearing away of the ozone is caused by human activities; activities that are somewhat
indulgent, careless and irresponsible. Thus, this erosion of the ozone presents an imminent threat to civilization and has lead
to severe health and environmental issues.
ultraviolet radiations. These radiations break apart oxygen molecules into separate oxygen atoms. When these atoms become
attached to some other oxygen molecules, ozone is formed. It collects in a layer from 19 to 35 km (known as the stratosphere,
whereas the lower part of the atmosphere is known as the troposhere). Scientists have been monitoring concentrations of
ozone in the upper stratosphere since evidence was found over Antarctica in the mid-1970's.
life on earth by filtering some of the sun’s powerful radiation. Besides acting as a canopy from strong radiation, the ozone also
acts as a purifier. Louis Vaczek highlighted in his book Enjoyment of Chemistry that ozone “destroys germs” and for this
reason is used to purify water and to clean public places. Also it combines much more rapidly with other substances and is
also used as a bleach for many common substances, such as flour, wax, and cloth.
release chlorine atoms. These chlorine atoms then help to break down ozone into ordinary oxygen, reducing the amount and
effectiveness of ozone in our atmosphere. Intensive and careful research done by Mario Molino, a postdoctoral student
revealed that just one chlorine atom could, through a chain of reactions, destroy thousands of ozone molecules. The damage
to Earth's shield's depends on how many CFC molecules is released every year. Each year these molecules drift upwards and
each of these CFC molecule takes between six and eight years to reach the stratosphere.
conditioning and refrigeration, as propellants for aerosol sprays, as blowing agents for foams, as cleaning agents in the
manufacture of electronic parts, including computer chips, and also as expansion agents. These very substances are
responsible for the present global ozone depletion. Who are responsible for CFC's in the atmosphere? The very manufacturers
of these substances and those who indulgently misuse these substances. Indeed, human activites has led and continues to
lead to depletion. In fact, the creation of the supersonic transport(SST), a passenger aircraft that was designed to cruise the
stratosphere sparked the first burst of atmospheric research. In 1971 Dr. James McDonald, an atmospheric physicist at the
University of Arizona, testified before a congressional subcommittee that flights by hundreds of SST's would destroy ozone,
thereby allowing more ultraviolet radiation to reach earth's surface and causing an increase in human skin cancers.
depletion through industrialization. The fact that they are useful often causes a compromise, allowing producers to overlook
the damage it causes and focus on the income it provides. In an effort to stop the manufacture of CFC's, the Berkeley council
aimed at removing the styrofoam containers made with chlorofluorocarbons "that keep the McDLT's hot side hot and cold side
cold." This occurred in 1988. This sparked a great deal of controversy because it obviously meant that companies using these
products would either have to substitute them, and possibly pay more for the substitutes. Joseph Bow, spokesman for the
Foodservice Packaging Institutes said, "The amount of CFC's we release is basically insignificant." However, as it later turned
out, when companies learned that McDonalds discontinued the use of the CFC containers, they did the same. Regardless of
how significant or insignificant the removal of certain CFC substances may seem, if cooperation is coupled with a desire to
save the ozone, these products will be completely removed and more concrete laws will be enforced to deal with this issue.
Afterall, it is not just a national issue but an international issue.
has not achieved it's goal. The amount of ozone has depleted tremendously since then, we are now in the year 2003, and face
the very same ordeal-trying to reach an agreement that will be adhered to internationally. Two revisions of this agreement
have been made in the light of advances in scientific understanding, the lastest being in 1992. It was anticipated that these
limitations would lead to a recovery of the ozone layer within 50 years of 2000, the World Meterological Organization estimated
2045 (WMO reports #25,#37), but recent investigations suggest that the problems is perphaps on a much larger scale than
expected. An article published by Year in Science in 2002 boldly declared "That Ozone Hole? Never Mind". The approach to
such a serious issue is evidently one of indifference. It mainly stated that the ozone will start the recovery process as soon as
2015. What about the CFC's that are still being released in the atmosphere?. Will the ozone recover, in spite of the increase in
industrialization and decrease in cooperation for a concrete agreement? These questions certainly make this revelation feeble.
More action needs to be taken to save the ozone, and very soon.
destroyed. In fact, a thinning usually occur for a few weeks in September to mid-November, during the Antarctic spring.
However, recent studies demonstrate that this phenomenon has taken a "swerve". While it is a natural process, such rapid
depletion is unprecedented. Depletion of the ozone has now created a hole in the ozone layer. Indeed, the whole had been
around since the mid-1970s and getting larger every year since the 1986, when it appeared to decrease by 10%. According to
a satellite vision in 1991 taken by Nasa,the total amount of ozone over a station in Antarctica where people were living and
working was shown to have decreased by about third between the mid-1980's. Measurement from this satellite showed that
depletion occured over all of this continent. By the early 1990's the amounts of ozone over Antarctica were about half those of
the 1970's. In fact, this depletion is rapid and devasting even as we speak. Scientists from New Zealand recently reported that
the hole in the Antarctic has for the first time stretched over a city. During two days in early September of the year 2002 the
hole extended over the southern Chilean city of Punta Arenas, exposing locals to high levels of ultraviolet radiation. Also citing
data from NASA, scientists said that the ozone hole now covered 11.4 million sqare miles (29.6 million square kilometers)-the
biggest it has ever been.
elsewhere. The Ozone Trends Panel, an international group of 130 scientists, whose work was coordinated by NASA, began
to investigate the matter in 1986. The scientists analyzed data from all over the world, and from both Dobson
spectrophotometers and ozone-measuring devices on satellites called total ozone mapping spectrometers(TOMS). In March
1988, the panel announced it's findings. It reported losses of ozone all over the world, including the Northern Hemisphere
, between 1978 and 1985. The ozone shield was thinning over China, Japan, Europe, what was then called the Soviet Union,
Canada, and the United States. The losses, up to 6 percent a year, were greatest in the winter time but occured in all seasons.
The panel's report stated that more ultraviolet ligth was reaching earth than at any time in modern history. They also concluded
that pollution was probabaly blocking some of it, but the increased UV-B radiation would cause more skin cancers and other
harm.
Cancer, an ailment (disease) that affects thousands of Americans and people of other nations, and has lead to the death of
various people. In fact, cancer is one of our society's most serious problems, second only to heart disease as a cause of death
in the United States. More precisely, upwards of 500,000 new cases are diagnosed each year: one in seven Americans will get
the disease in his or her lifetime, even with no depletion of the ozone layer. This is terrifying because it makes one wonder
about consequences with the ozone now decreasing. Therefore making it one of the greatest threats that humanity face.
Statistics demonstrate that the global deterioration of the ozone layer is responsible for the most notably increase in the rate oof
skin cancer. Just how is this possible? A brief article issued by Science News, tells how this happens based on an experiment
done by Jan C. Van der Leun and his colleague Frank R. de Gruiji. The investigation was done with mice, and the culmination
proves that it takes about the same UV dose to induce sunburn in humans as it does mice. De Gruijl explained that "What's
vulnerable to carcinogenic transformation however, is not the surface skin, but the proliferating cells below". They tuned data
on UV's skin carcinogenicity--its action spectrum--in this to account for the longer path UV rays must traverse through human
skin. The results for long-wavelength(A) portion of the UV spectrum now indicates that UV-A offers five times the human
cancer risk seen in the mouse action spectrum. The new human action spectrum now indicates that the longer UV-A
exposures needed to create a tan would pose about the same cancer risk as a UV-B tanning dose. Therefore fortifying the
evidence that the thinner the ozone, the more ultraviolet radiation is allowed to reached Earth and ultimately affect it's
inhabitants, since there is less protection. In fact, the new human UV action spectrum indicates that when it comes to a
permanent thinning of Earth's ozone, each 1 percent drop in stratospheric ozone could increase the incidence of
nonmelanoma skin cancers by 2 percent.
shows that the excess ultraviolet rays from sunlight kills amphibians (salamanders etc.) in Oregon's Cascade Mountains. (The
location also proves that the ozone depletion is not solely over Antartica, but affects various areas of the world, making it an
international crisis and concern). This can be explained by the fact that many species of toads, frogs, and salamaders lay eggs
that are usually unprotected from increased ultraviolet light. These eggs are laid in shallow water and are not protected by
shells. For those wondering, can ultraviolet rays actually affect shallow water? Yes, in fact, when biologist used a filter to block
UV-B rays from the frog and toad eggs, the survival of the young in the eggs increased dramatically. All over the world,
populations of frogs and toads have been declining, especially in mountain areas, and increased ultraviolet light caused by an
increase in the amount of ozone depletion is likely the cause.
cataract-and you certainly can't put sunscreen in your eyes!. These wavelengths can cause damage to the immune system
and make the body more vulnerable to disease. Wildlife in Antarctica are severely affected, since the depletion is more
concentrated in that continent. While penguins and seals are shielded from UV-B rays by feathers or fur, and penguin and
seabirds eggs are opaque and thus protected from ultraviolet light, powerful wavelengths can damage their eyes, though.
some of the effects of a rapidly decreasing ozone. Hundreds of millions of people are now piosed on the threshold of a skin
cancer epidemic, and the world's ecosystem are undergoing profound damage. Animals can also be harmed in indirect ways:
through their food supply. Environmental groups and biologists have warned that ultraviolet light might harm phytoplankton,
the microscopic drifting algae that are the first link in many Antarctic food chains. Small crustaceans called krill are the next
link in many food chains, and fish, penguins, seabirds, seals, and certain kinds of whales feed on krill. In 1989 Dr. Sayed El-
Sayed, a plant ecologist at Texas A&M University, said "Krill depend on Phytoplankton, and if anything happens to krill, the
whole ecosystem will collapse". This can be seen in any ecosystem. It is a known fact that even a minor disturbance in a
natural ecoystem can result in devastating consequences. Therefore, can you imagine what will happen if this depletion
continues at such a rapid pace?. These phytoplankton can be harmed in two major ways. First, their food-making process-
photosynthesis- could be diminished: second, UV-B rays could damage their genetic material and prevent them from
reproducing. Scientists have found that ultraviolet rays penetrate sixty feet deep in clear water on a cloudless day but are most
powerful in the top few feet- the depth to which phytoplankton rise, because they need certain wavelengths of solar energy
found close to the surface for food making.
and unmonitored human activity can actually have on the environment. This depletion has lead to environment issues, such as
loss of animals and health related issues, especially those tied to an increase in the cancer rate. It also poses grave questions
about agriculture and the impact the world would experience if permanent depletion is a result of the rapid decrease that we
are experiencing. Little wonder environmentalists describe it as a "crime against humanity". In order to preserve what is left of
the ozone, we as a nation, as an international body must be motivated to take action that will gave us and our posterity a
chance to live in a fearless environment, knowing that the ozone layer is capable of performing the role for which it was
formed. In 1933, Dr. Charles Abbot, of the Smithsonian Institution in Washington, D.C., said,"It is astonishing and even
terrifying to contemplate this narrow margin of safety on which our lives thus depend. Were this trifling quantity of atmosphere
ozone removed, we should all perish
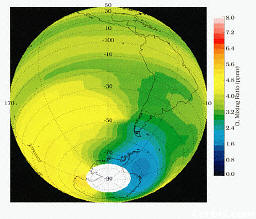
levels of ozone in the atmosphere of the Southern Hemsiphere, as measurec by NASA's HALOE experiment.
Davis, A. Donald. “Saving the Earth". Drug &Cosmetic Industry. Advanstar Communications Inc, 1995. 20
.“Estimating Uv’s Human Cancer Risk”. Science News. New York: Science Service, Inc. 1994. 255
.Lait, Matt. “Holding The Foam To Save The Ozone”. Washington Post. Washington,D.C. Feb 16th, 1988. p. A3
.Laurence, Pringle. Vanishing Ozone: Protecting Earth From The Ultraviolet Radiation. New York: Morrow Junior Books, c1995. 1-64
."Ozone Loss Tied to Animal Deaths". The New York Times. New York: MIT Press Journals,2000. 14.
“Ozone still Growing”. Ecologist. New York: MIT Press Journals, 2002. 8 Singer, S. Fred and Crandal, Candace. “Assessing the Threat to the Ozone.” Consumers’ Research. July 1987. 11-14 Townsend, Mark. “Chilling Evidence”. Geographical. New York: Campion Interactive Publishing Ltd, 2002. 54-57 Vaczek, Louis. Enjoyment of Chemistry Winters, Jeffrey. “That Ozone Hole? Never Mind”. Year In Science. 2002, p. 27