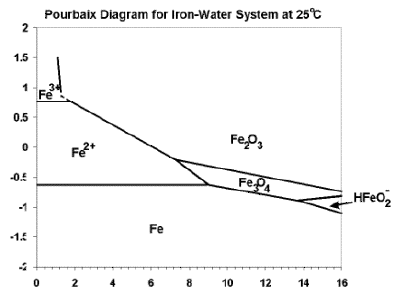
Through
the use of thermodynamic theory (the Nernst equation), so-called Pourbaix
diagrams can be constructed. These diagrams show the thermodynamic stability of
species as a function of potential and pH. Although many basic assumptions must
be considered in their derivation, such diagrams can provide valuable
information in the study of corrosion phenomena. The diagram below represents a
simplified version of the Pourbaix diagram for the iron-water system at ambient
temperature. For the diagram shown, only anhydrous oxide species were considered
and not all of the possible thermodynamic species are shown.

Some
limitations of such diagrams include: