Copyright © Dr. Shiwei Guan, all rights reserved.
![]()
Copyright © Dr. Shiwei Guan, all rights reserved.
SYNERGISTIC PROTECTION AGAINST MICROBIOLOGICALLY INFLUENCED CORROSION USING A 100% SOLIDS POLYURETHANE INCORPORATED WITH ANTI-MICROBIAL AGENTS
Shiwei Guan, Ph.D.
Director, R & D
Madison Chemical Industrial Inc., 490 McGeachie Drive, Milton, Ontario L9T 3Y5 Canada
E-mail: sguan@madisonchemical.com
ABSTRACT
This paper summarizes various mechanisms of microbiologically influenced corrosion (MIC) in municipal wastewater applications as it relates to steel, ductile iron, and reinforced concrete substrates. A novel method of using a 100% solids polyurethane coating system incorporated with anti-microbial agents is presented and its synergistic benefits are discussed.
1. INTRODUCTION
Microbiologically influenced corrosion or microbiologically induced corrosion (MIC), refers to corrosion and ensuing loss of metal or concrete caused by biological organisms. MIC can occur in any aqueous environment, and it is now a commonly occurring phenomenon because of the omnipresent nature of microbes, adequate nutrients, and corrosive byproducts in today's industrial and municipal processes. Protection of structures against microbiologically influenced corrosion has therefore become very critical in many industries including municipal pipeline, marine, storage vessels, sewage treatment facilities and so on.
In municipal wastewater collection and treatment, it has become painfully evident to many design engineers and utilities that they are facing extremely aggressive corrosion conditions. Corrosion problems in municipal wastewater treatment and collection systems are quite different than those typically found in industrial systems. In the latter, corrosion results from direct aggression by the chemicals and their reaction products present in various discharge streams serviced by the system. Therefore, corrosion in those industrial systems usually occurs below the water line and tends to be predictable in nature. In municipal systems, however, the most common corrosion encountered is above the water line and is typically microbiologically induced. Other chemicals in the waste stream may also cause corrosion below the water line. Moreover, the rate of attack is difficult to predict.
By providing a barrier between wastewater and various substrates such as steel, ductile iron, and concrete, protective coatings are the most common solutions adapted by many municipalities around the world against microbiologically influenced corrosion. The unpredictability in rate of attack referred to above means that virtually every municipality has reasons to be concerned. For metallic structures or pipes, the most common coatings systems used are coal tar epoxy, polyethylene, ceramic epoxy, and more recently 100% solids polyurethane.1 For concrete substrates, PVC (Polyvinyl Chloride) and coal tar epoxy have been used extensively since the 1940s. 2,3 There is a great challenge, however, to provide a more economical and also a more reliable coating particularly for concrete substrates.4
A relative newcomer to the wastewater industry, 100% solids polyurethane has been extensively used since the 1970s. Because of their impermeability, resistance to microbial attack, excellent adhesion and abrasion resistance, flexibility, and unlimited film build, some 100% solids polyurethanes have accumulated a significant track record of excellent success in the protection of steel, ductile iron, and concrete surface exposed to MIC in wastewater applications. 1,4
In addition to the approach of using protective coatings, various efforts have also been made in order to reduce or eliminate the source of microbiologically influenced corrosion. These efforts include environment alteration, proper design of structures and pipelines, cathodic protection, and the use of anti-microbial agents.
The purpose of this paper is to summarize various mechanisms of microbiologically influenced corrosion (MIC) in municipal wastewater applications as it relates to steel, ductile iron, and reinforced concrete substrates. A novel method of using a 100% solids polyurethane coating system incorporated with anti-microbial agents is presented and its synergistic benefits are discussed.
2. MECHANISMS OF MIC IN WASTEWATER APPLICATIONS
2.1 GENERAL
Microbiologically induced corrosion (MIC) results from the corrosive secretions of microorganisms. Biological organisms fall under two groups based on the type of corrosion they engender: (a) anaerobic corrosion and (b) aerobic corrosion. Sulfate reducing bacteria (SRB) from the genera desulfovibrio are a typical example of anaerobic MIC. In municipal wastewater systems, the SRBs responsible for anaerobic MIC are primarily Thiobacilli, which thrive on the sewage environment. System structures are rarely completely filled with sewage; thus, ample space exists above the water line for bacterial growth and gaseous products from the decomposition of sewage to collect. Due to their extremely rapid rate of reproduction, enormous colonies of bacteria are produced in a short time, resulting in a large potential source of corrosive media.
The MIC mechanism begins with the fermentation of raw sewage that generates methane and H2S gases. When the wastewater stream is anaerobic, SRBs existing in the slim layer in the submerged area convert the naturally occurring sulfates in the wastewater into H2S. The hydrogen sulfide, methane, and carbon dioxide (CO2) react with water vapor to produce a mild acidic condition which condenses on the structure/pipe surface above the water line. This process lowers the pH of the substrate surfaces to levels favorable for the growth of the Thiobacilli bacteria. The Thiobacilli oxidize H2S and other sulfur bearing materials in the wastewater during their respiration and secrete sulfuric acid as a waste. In turn, each strain of bacteria lowers the pH of the substrate surface to an optimum range for another strain to lodge and reproduce. This process is repeated throughout the chain for all of the Thiobacilli strains involved, creating a very acidic environment, thereby encouraging rapid corrosion.
2.2 fACTORS INFLUENCING HYDROGEN SULPHIDE PRODUCTION
When there are problems in drainage systems caused by H2S, the following factors are usually the main influences in the production of hydrogen sulfide that may lead to the formation of sulfuric acid in sewers.
a. Dissolved sulphide
The sulfide concentration is the limiting factor in the release of hydrogen sulfide to the sewer walls so that corrosion may occur. If metals are present in the sewage stream, a small amount of sulfide is immobilized to form insoluble metal salts. The amount varies from 0.1 to 0.3 milligrams per liter.
b. pH
The pH influences dissociation of the sulfide ion species in the sewer. At a pH of 6, more than 90% of the dissolved sulfide is hydrogen sulfide. At a pH of 8, less than 10% is in the form of hydrogen sulfide.
c. BIOLOGICAL OXYGEN DEMAND (BOD) AND TEMPERATURE
Temperatures above 15oC (59oF) may contribute to the generation of hydrogen sulfide if all other conditions of sulfide generation are present.
BOD is a measure of the oxygen depletion by the decomposition and mineralization of organic matter. In a sewer system, the conversion of sulfates to sulfide requires energy. The BOD determination is a measure of the energy within the system that will facilitate this conversion. The BOD usually occurs over a 5-day period and has thus become known as the 5-day BOD.
d. VELOCITY
Velocity affects the rate of oxygen absorption, the release of hydrogen sulfide to the atmosphere, and the build up of solids. The minimum velocity of the sewer stream should be between 0.61 and 1.07 meters per second to keep solids in suspension. If the velocity causes turbulent flow conditions, increased oxygen may be absorbed into the wastewater, but hydrogen sulfide in wastewater will also be released to the atmosphere. The released hydrogen sulfide may cause corrosion to the wall of the wastewater pipe.
e. JUNCTIONS
Junctions are important because the wastewater from tributaries may contain high concentrations of sulfide, lower pH, high BODs, and higher temperatures. All of these factors may affect the hydrogen sulfide production in the main sewer line. Junctions may also affect the type of flow where they enter the main. If the flow is turbulent, more oxygen may be absorbed into the wastewater, or more hydrogen sulfide may be released into the atmosphere. Since the effects of corrosion outweigh the increase in oxygen absorption, the junctions should enter the main in a manner that reduces turbulence.
f. FORCE MAINS AND SIPHONS
Special junctions like force mains and siphons, have a similar effect on the quality of the wastewater stream, as do regular junctions. Force mains and siphons may flow at low velocities, or intermittently, allowing the increase of sulfide. Force mains usually flow full, which also facilitates the build-up of sulfides due to the anaerobic conditions in the force main. When force mains and siphons enter the main sewer, the higher concentration of sulfide may cause problems further downstream.
g. VENTILATION
Ventilation is not an effective measure to reduce the corrosion of wastewater pipe because it is difficult to prevent condensation on the walls of the pipe due to temperature variations. The hydrogen sulfide is oxidized in the aerobic layer on the wall of the pipe to form sulfuric acid, which may corrode the pipe as it trickles down the wall of the pipe.
If velocities of 0.61 meters per second and oxygen level of 1 milligram per liter and temperatures less than 15oC (59 oF) can be achieved, corrosion in sanitary sewers will not be a problem at any time.
Accumulation of solids could be a problem during the three warmest months of the year. During these months, the temperature is sufficiently high to have sewer water temperatures above 15oC (59 oF). The elevated temperatures would also decrease the dissolved oxygen. Dissolved oxygen is inversely proportional to the temperature of the water. If effective BOD levels are less than 600 milligrams per liter and the effective slope is 0.2 % and flow is 0.085 cubic meters per second, sulfide concentrations will not increase sufficiently to become a problem.
2.3 STEEL AND DUCTILE IRON
A number of metals, such as structural steels, mild carbon steels, ductile iron etc., tend to corrode generally over the entire surface in the absence of crevices or galvanic effects. In such cases, corrosion is determined by the rate at which dissolved oxygen can be delivered to the metal surface. Biological organisms present in the aqueous medium often have the potential to increase or decrease oxygen transport to the surface; consequently, these organisms play a role in increasing or decreasing general corrosion. Most MIC, however, manifests as localized corrosion because most organisms do not form in a continuous film on the metal surface. Microscopic organisms tend to settle on metal surfaces in the form of discrete colonies or at least spotty, rather than continuous films.
The classic mechanism for MIC of steel and iron was proposed by Von Wolzgen Kuhr in 1934.6 This mechanism is based on the idea that the rate-limiting step in corrosion is the dissociation of hydrogen from the cathodic site. It is thought that sulfate-reducing bacteria (SRB) consume hydrogen through the action of their hydrogenase enzymes, and thus "depolarize" the cathode, accelerating corrosion. Some investigators still believe that this mechanism is the important one for MIC of iron and steels, despite the fact that numerous experiments using SRB in pure culture gave corrosion rates far less than those seen at field sites and less than those measured in experiments using MIC communities.
Most coatings systems used for protecting a metal from MIC have been working well. Even after being coated with polymeric coatings, however, a metal may still be open to biologically influenced attack either through biodeterioration of the coating or through MIC of the underlying metal. Microorganisms are frequently implicated both in the corrosion of metals and in the deterioration of complex polymeric coatings. Deterioration may involve blistering, delamination, and changes in porosity, selective leaching of material components and changes in material properties by contamination from microbial metabolites. Corrosion of the substrate metal may be directly or indirectly influenced by the presence of organisms.7
2.4 CONCRETE
Wherever there is a serious septic sewage problem, corrosion of concrete surfaces occurs. Although having an initial alkalinity as high as a pH of 13 due to the formation of lime in the hydration of dicalcium and tricalcium silicates (Portland cement components), concrete surfaces could have a pH as low as 0.6 because of microbiologically induced attack. The sulfuric acid content on the exposed areas has been measured to as high as 5%5 or even 10%.8 This concentration of sulfuric acid corresponds to a pH value less than 1.0. The sulfuric acid generated due to MIC directly attacks the underlying substrate and causes destruction of the infrastructure. In many instances, microbiologically induced corrosion has damaged concrete structures to the point where major rehabilitation was required in as few as four years, or total collapse occurred within six years.
Many coatings have been applied to such concrete sewer structures and piping. Almost all have resulted in a complete failure.8 There are a few coatings systems that would withstand sulfuric acid, however, the actual coating of concrete is a more complicated and difficult procedure than providing the same type of protection to steel. Steel is a smooth, impervious, uniform, dense surface, while concrete is soft, brittle, subject to cracking, filled with air and water pockets, porous, water permeable, and chemically active. A coating over concrete surfaces is therefore subject to imperfections, and even a minute imperfection will cause the concrete to disintegrate under the coating.
3. protection against mic using a 100% solids polyurethane
For protection against microbiologically influenced corrosion, it is essential to achieve a continuous protective coating barrier that will resist highly aggressive chemicals such as sulfuric acid. Also important are adhesion, abrasion resistance, cathodic disbondment resistance, impact resistance, and low permeability.
Since 1993, a 100% solids polyurethane coating, developed by the author at Madison Chemical, has been successfully used as an internal lining system for hundreds of miles of steel, ductile iron, and concrete pipes serviced in North American wastewater industry. In addition to its excellent handling characteristics such as cold temperature cure, fast setting and unlimited film build, zero VOC's, and non-leaching, the 100% solids polyurethane coating has the performance characteristics as follows.
Table 1 Performance Characteristics of The 100% Solids Polyurethane Coating
Characteristics |
Method |
Results |
| Chemical resistance to 20% H2SO4 | ASTM D714, 100,000 hours continuous immersion, coated on steel and ductile iron | No change, No blisters |
| Adhesion to steel | ASTM D 4541 |
2000 p.s.i. (glue failure) |
| Adhesion to ductile iron | ASTM D4541 |
2600 p.s.i. (cohesive failure) |
| Adhesion to concrete | ASTM D4541 |
Exceeds cohesive strength of concrete (500 p.s.i.) |
| Abrasion resistance | ASTM D4060, CS17, 1 kg, 1000 cycles |
78 mg loss |
| Cathodic disbondment | ASTM G95 |
9.9 mm radius |
| Flexibility and resistance to cracking | ASTM D522 |
180o over a 2" mandrill (12% elongation) |
Impact resistance |
ASTM D2794 at 40 mils |
110 in-lbs. |
Permeability |
ASTM D1653 |
(non-breathing) 0.005 perm-inches |
Salt spray resistance |
ASTM D714, 1000 hours, coated on steel and ductile iron | 0 under film corrosion |
It has been proven that a 40 mils thick film of the 100% solids polyurethane coating can provide a totally holiday free layer (under holiday testing of 100 volts per mil or higher) on steel and ductile iron substrates. The above performance testing results merit the outstanding protection of these substrates against microbiologically influenced corrosion.
There is no systematic method for evaluating the performance of a coating system for a concrete substrate under both wet and dry conditions. However, the current environment of fiscal restraint and fiscal responsibility has encouraged, if not forced, municipalities to design their wastewater structures for maximum longevity. Full testing programs have therefore been developed by some municipalities to evaluate all available coatings systems for concrete and clay brick facilities. Among these programs one that should be particularly mentioned is the study conducted by the Greater Houston Wastewater Program, City of Houston.9
The testing program used full scale pressure chambers (hydrostatic tests), as shown in Figure 1, to evaluate the application and performance of coating materials on concrete substrate under a hydrostatic pressure of over 10 m (32 ft.) of water simulating the ground water conditions. Coated concrete and clay bricks with man-made holidays to simulate pinholes of the coating film in real application, as shown in Figure 2, were used to study the chemical resistance under an acidic environment (ASTM G20-88). To quantify bonding strength between the coatings and substrates two ASTM standard testing methods were used (ASTM D4541 and ASTM C321).
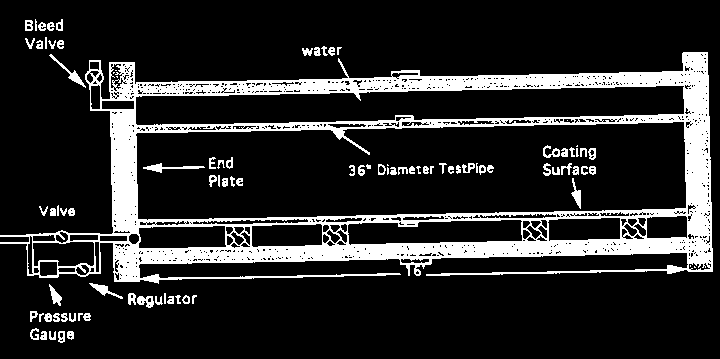
Figure 1 Hydrostatic pressure test chamber used for evaluating the application and performance of coatings on concrete surface 9
Results based upon the full-scale test (five months) and laboratory tests (six to ten months) of the 100% solids polyurethane coating are summarized as follows:
Application: The 100% solids polyurethane coating was applied successfully under dry and wet (hydrostatic test) conditions respectively. No defects (blistering, cracking, discoloration, spalling, sticking to the finger after 48 hours of application, scratch-off) were observed on either dry or wet concrete surfaces.
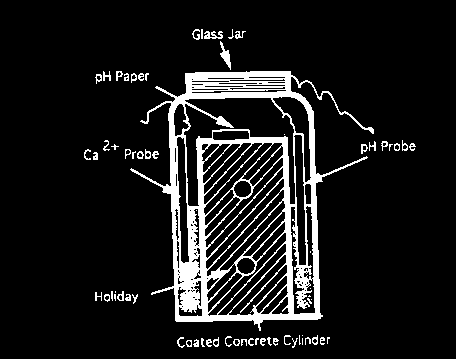
Figure 2 Chemical test with holiday for coated concrete specimens (modified ASTM G 20) 9
Performance: Coatings were tested under a hydrostatic pressure of 105 kPa (15 p.s.i.) over a period of five months. The coatings were inspected on a regular basis to identify any visible defects for (a) overall condition (b) surface texture (c) blistering (d) cracking (e) change in color and (f) quality of finish. The 100% solids polyurethane coating passed the performance test in all the above categories.
Holiday Test - Chemical Resistance: Coated concrete cylinders and clays were tested for over a period of six months with and without man-made holidays in water, 3% sulfuric acid and 30% sulfuric acid solutions. The holidays had a diameter of 1/4 inch, 1/2 inch, and 1/8 inch and a depth of 1/4 inch, 1/2 inch, and 1/8 inch, respectively. The 100% solids polyurethane coating passed the vapor phase and liquid phase tests with no visible blistering, discoloration, or cracking.
Bond Strength: Bond strengths of the coatings were determined according to ASTM D4541 and ASTM C321 over a period of ten and seven months respectively. For the dry concrete substrates coated with the 100% solids polyurethane coating, all the failures were associated with the failure of concrete in direct tension. Hence the bonding strength of the coating is higher than the concrete strength.
It is interesting to note that all the concrete substrates coated with the 100% solids polyurethane with the intentional holiday passed the 30% sulfuric acid test after six months of immersion. This result is believed to be due to a mechanism of local neutralization within the holiday. Within the holiday, sulfuric acid due to MIC first reacts with the exposed concrete surface. Because of the high alkalinity of the concrete, however, the local pH value will increase, making reacted deposits thermodynamically stable within the holiday. If the coating layer around the holiday has a good bond strength to the substrate, the size of the holiday will remain unchanged. The insoluble and stable deposits are then acting as a physical barrier over the reacted concrete surface, preventing it from further corrosion.
Another long-term in-house test also confirms the beneficial effect of the 100% solids polyurethane coating on concrete when small intentional holidays are present. A plastic cylinder was attached to three concrete pipe blocks respectively and sealed with a two component and chemical resistant polyurethane touch-up/repair material. One concrete pipe block was non-coated and two were coated with the 100% solids polyurethane. A 6-mm (0.25 inch) diameter holiday was drilled through the coating to expose the concrete substrate. A 30% sulfuric acid solution was filled into the plastic cylinder on the bare concrete sample and one coated sample. The other coated sample was filled with a 3% sulfuric acid solution. After only 45 days, the bare concrete sample corroded all the way underneath the polyurethane sealer and caused the leakage of the 30% sulfuric acid solution. The two-coated samples had not shown any leakage even after 21 months, neither displaying any softness nor damage. The intentional holidays on the two samples were blocked with deposits, acting as a barrier together with the polyurethane coating. The average disbonding radius of the coating from the initial holiday was 15 mm and 18 mm for 3% and 30% sulfuric acids, respectively.
There are many factors that could affect the protection due to the "local neutralization" phenomenon. Firstly, if the pH of the surrounding area is too low, the local neutralization could disappear over a long period of time as acidic species diffuse from the bulk into the local holiday. Secondly, the excellent bond strength (adhesion) of a coating to the concrete substrate is also very critical, in order to prevent the size of the holiday from becoming larger.
4. protection using the 100% solids polyurethane Coating incorporated with anti-microbial agents
A modified version of the above 100% solids polyurethane system has been made. The modification is the addition of 0.1-0.5% of an anti-microbial agent into the existing coating formulation. The anti-microbial agent used is a non-toxic, stable to 125oC (256oF), UV stable material, containing no phenols, tin, heavy metals, lead, mercury, or formaldehyde. The anti-microbial agent acts as microbial 'spears' that pierce the Thiobacillus cells, inhibiting the growth of the cells. The anti-microbial agent is also chemically reacted with the coating resins and incorproated into the polymeric. This enables its permanent presence across the whole coating film and causes no-leaching to the environment.
A testing program has been set up to evaluate how the modified 100% solids polyurethane coating and coated surface is protected with the anti-microbial agent. Three most common microorganisms found in wastewater applications were tested: Thiobacillus thiooxidans, Thiobacillus thiooparus, and Thiobacillus denitrificants.
After pH adjustment with carbon dioxide, a 0.2-milliliter aliquot of a bacterial suspension of the Thiobacillus species was aseptically pipetted evenly onto the upper surface of each disc and incubated at 25oC for 24 hours.
The upper surface of the discs were washed with 10 ml of ATCC medium 238 for Thiobacillus thiooxidans, and 10 ml of ATCC medium 290 for Thiobacillus thiooparus and Thiobacillus denitrificants. The cell suspension was then collected into a sterile container and ten-fold serial dilutions of the bacterial suspension were made in the appropriate medium to obtain viable counts using a modified NETAC method. Four replicates were made per set.
As a comparison, control samples (the unmodified 100% solids polyurethane coating) were also used to test the effect of the coating on the three microorganisms. However, as expected, no anti-microbial activity was found.
The percentage of bacterial reduction of the anti-microbial agent from the modified 100% solids polyurethane coating is summarized in Table 2. It clearly suggests that the anti-microbial agent will remain active after incorporation into the 100% solids polyurethane, as 99.9% of the tested organisms were killed.
Table 2. % Reduction of Thiobacillus microorganisms by the anti-microbial agent after incorporated into the 100% solids polyurethane coating10
Microorganisms |
Viable count / ml. (initial) |
Viable count / ml. (recovered from water) |
% Reduction |
Thiobacillus thiooxidans |
1 x 105 |
1 x 101 |
99.99 |
Thiobacillus denitrificans |
1 x 106 |
1 x 102 |
99.99 |
Thiobacillus thioparus |
1 x 105 |
1 x 102 |
99.99 |
The second set of tests is designed to determine if the anti-microbial agent is active under simulated conditions of constant water flow. Anti-microbial activity was tested on surfaces of samples previously subjected to 110 hours of constant running tap-water immersion. The tests follow the same testing procedures as described above. Percentage of reduction of the microorganisms is shown in Table 3.
Table 3. % Reduction of Thiobacillus microorganisms by the anti-microbial agent in the 100% solids polyurethane coatings previously subjected to running water immersion10
Microorganisms |
Viable count / ml. (initial) |
Viable count / ml. (recovered from water) |
% Reduction |
Thiobacillus thiooxidans |
1 x 106 |
1 x 101 |
99.999 |
Thiobacillus denitrificans |
1 x 106 |
1 x 102 |
99.99 |
Thiobacillus thioparus |
1 x 106 |
1 x 102 |
99.99 |
The third test is designed to determine if the anti-microbial agent is present and active throughout the material or simply at the surface of the 100% solids polyurethane coating. A "V" shaped section was removed from each sample of 1/2-inch thick coating free film to completely expose the interior of the coating cross section. A 0.2-ml aliquot of a bacterial suspension of the different Thiobacillus species was aseptically pipetted onto the newly exposed cross section area and incubated at 25 degrees centigrade for 24 hours. The test samples were then treated as described for the surface samples. Percentage of reduction of the microorganisms is shown in Table 4.
Table 4. % Reduction of Thiobacillus microorganisms by the anti-microbial agent in the cross sections of the 100% solids polyurethane coatings10
Microorganisms |
Viable count / ml. (initial) |
Viable count / ml. (recovered from water) |
% Reduction |
Thiobacillus thiooxidans |
1 x 106 |
1 x 101 |
99.999 |
Thiobacillus denitrificans |
1 x 106 |
1 x 101 |
99.999 |
Thiobacillus thioparus |
1 x 106 |
1 x 101 |
99.999 |
Results of the above three tests were so unique that three other microorganisms were then introduced into the test program. These three microorganisms are: Enterobacter aerogenes, a gram negative coliform; Pseudomonas mendocina, a gram negative rod considered to be resistant to anti-bacterial chemicals, and Arthrobacter citreus, a gram positive rod. They are not normally present in wastewater applications, but are regarded as very resilient microorganisms. The test procedure was essentially the same as with the Thiobacillus species with the modified 100% solids polyurethane coating. The results are shown in Table 5.
Table 5. % Reduction of three highly resistant microorganisms by the anti-microbial agent in the cross sections of the 100% solids polyurethane coatings10
Microorganisms |
Viable count / ml. (initial) |
Viable count / ml. (recovered from water) |
% Reduction |
Enterobacter aerogenes |
1 x 107 |
00.00 |
100 |
Pseudomonas mendocina |
1 x 106 |
00.00 |
100 |
Arthrobacter citreus |
1 x 107 |
00.00 |
100 |
5. SUMMARY
Mechanisms of microbiologically influenced corrosion (MIC) in municipal wastewater applications, as it relates to steel, ductile iron, and reinforced concrete substrates, have been discussed.
The 100% solids polyurethane coating system will provide excellent protection against microbiologically influenced corrosion (MIC). By incorporating with an anti-microbial agent, the 100% solids polyurethane system and substrate surfaces coated with the modified coating will further be protected from the attack of microorganisms that are normally found in wastewater application.
References
S. Guan and H. Kennedy, "A Performance Evaluation of Internal Linings for Municipal Pipe", NACE International/Corrosion 96, Paper No. 482, Houston, 1996.
Ameron International, "T-Lock PVC Sheet Liner for Concrete Pipe and Structures R9-96", Brea, 1996.
TPC 12 (NACE International), "Coal Tar Epoxy Coating – A State of the Art Review", Houston, 1987.
Howard Kennedy, "Protection of Reinforced Concrete Pipe Against Microbial Induced Corrosion", NACE International/Corrosion 97, Paper No. 392, Houston, 1997.
A.M. Horton, "Special Protective Coatings and Linings for Ductile Iron Pipe", Advances in Underground Pipeline Engineering, ASCE, Bellevue, 1995, p. 750.
D. Pope and E. Morris, "Mechanisms of Microbiologically Induced Corrosion (MIC)", Materials Performance, Vol. 34, No. 5, NACE International, May 1995, p. 24.
D.B. Mitton, et al., "Microbially Influenced Corrosion of Polymer-Coated Metallic Substrates", 1st Mexican Symposium and 2nd International Workshop on METALLIC CORROSION, University of South Florida, 1997.
C.G. Munger, "Corrosion Prevention by Protective Coatings", NACE International, 1984, p.302.
C. Vipulanandan, et al., "Evaluating Madison Chemical Industries, Inc. Product for Coating Wastewater Concrete and Clay Brick Facilities in the City of Houston", Report No. CIGMAT/UH 96-5, December 96.
C.L. Baugh, Test Report # 970061, Custom Biologicals, Inc., August, 1998
SG/sg. Word 6.0, U:\DATA\GENERAL\DRAFT.LIT\55TECH\DEV130B.DOC